A simple cubic cell has an atom at each corner of a cube for a total of 8 atoms. However, the atoms are not inside the cube; in fact, only 1/8 of each atom is actually inside the cube.
So, there is only one atom inside the cube drawn on the right.
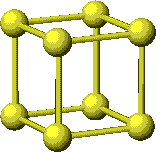
To determine the formula of a compound from its unit cell, one needs to know how many atoms are contained within the unit cell.
|
A simple cubic cell has an atom at each corner of a cube for a total of 8 atoms. However, the atoms are not inside the cube; in fact, only 1/8 of each atom is actually inside the cube. So, there is only one atom inside the cube drawn on the right. |
 |
To count atoms in the unit cell, count up all the atoms at each position. Then multiply by the fraction that is inside the unit cell.
1/8 of an atom that is on a corner is inside the unit cell.1/4 of an atom that is on an edge is inside the unit cell.
1/2 of an atom that is on a face is inside the unit cell.
Determine the formula of NaCl from the unit cell.
|
8 Cl- ions on corners |
|
|
|
6 Cl- ions on faces |
|
|
|
|
||
|
|
||
|
1 Na+ ion in the center |
|
|
|
12 Na+ ions on edges |
|
|
|
|
||